Home > Research Teams > Structure and Dynamics of Isolated Complex and Photoexcited Systems > Chirality and Spectroscopy > Spectrosccopy of cyclic dipeptides
Spectrosccopy of cyclic dipeptides
by - 3 August 2018 (modifié le 25 March 2020)
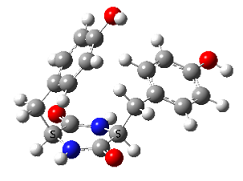
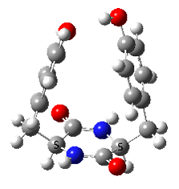
- Families of Tyr-Pro cyclo structures, LL on the left and LD on the right
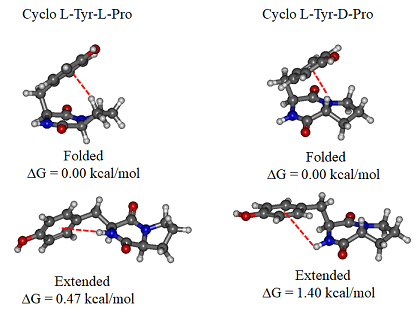
In contrast, very strong chirality dependence of the structure is observed for cyclo tyrosine-tyrosine, cyclo Tyr-Tyr. While both LL and LD diastereomers show the same “folded extended” structure as cyclo Phe-Phe, a structure involving a strong hydrogen bond between the hydroxyls of the two tyrosine residues is observed for LL only.
|
|
Publications
Conformational Study of the Jet-Cooled Diketopiperazine Peptide Cyclo Tyrosyl-Prolyl
A. Pérez-Mellor, I. Alata, V. Lepere, and A. Zehnacker
Journal of Physical Chemistry B, 2019, vol 123 (28), 6023-6033
Stereochemistry-dependent hydrogen bonds stabilise stacked conformations in jet-cooled cyclic dipeptides: (LD) vs. (LL) cyclo tyrosine–tyrosine
F. BenNasr, A. Pérez-Mellor, I. Alata, V. Lepere, N.-E. Jaïdane, and A. Zehnacker
Faraday Discussions, 2018, vol 212, 399-419
Chirality effects in the structures of jet-cooled bichromophoric dipeptides
A. Pérez-Mellor, I. Alata, V. Lepere, and A. Zehnacker
Journal of Molecular Spectroscopy, 2018, vol 349, 71-84
Vibrational circular dichroism of a 2,5‐diketopiperazine (DKP) peptide: Evidence for dimer formation in cyclo LL or LD diphenylalanine in the solid state
Ariel Pérez‐Mellor, Anne Zehnacker
Chirality, 2017, vol 29 (2), 89–96.